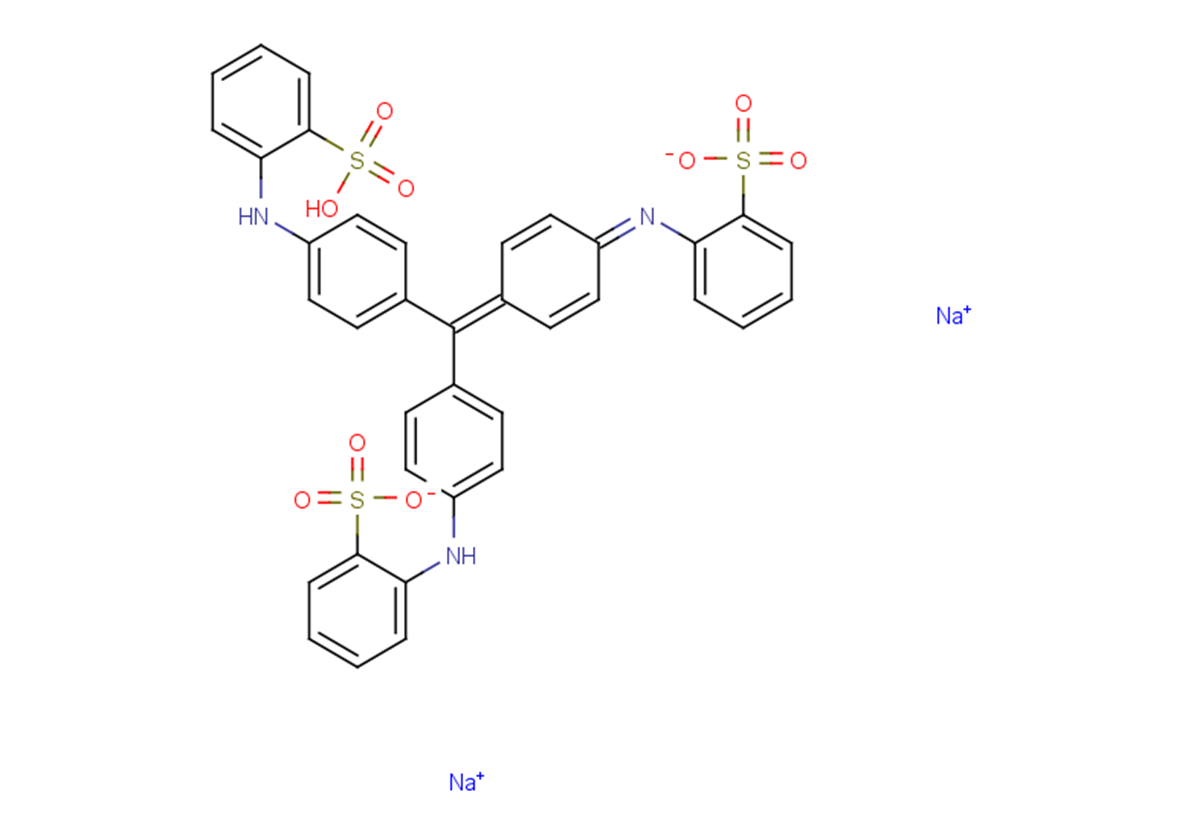
Methyl Blue
CAS No. 28983-56-4
Methyl Blue( —— )
Catalog No. M24207 CAS No. 28983-56-4
Methyl blue is a triaminotriphenylmethane dye. It is used as an antiseptic dye in the polychrome staining method and has applications in histological and microbiological staining solutions.
Purity : >98% (HPLC)
 COA
COA
 Datasheet
Datasheet
 HNMR
HNMR
 HPLC
HPLC
 MSDS
MSDS
 Handing Instructions
Handing Instructions
| Size | Price / USD | Stock | Quantity |
| 500MG | 26 | In Stock |


|
| 1G | Get Quote | In Stock |


|
Biological Information
-
Product NameMethyl Blue
-
NoteResearch use only, not for human use.
-
Brief DescriptionMethyl blue is a triaminotriphenylmethane dye. It is used as an antiseptic dye in the polychrome staining method and has applications in histological and microbiological staining solutions.
-
DescriptionMethyl blue is a triaminotriphenylmethane dye. It is used as an antiseptic dye in the polychrome staining method and has applications in histological and microbiological staining solutions.
-
In Vitro——
-
In Vivo——
-
Synonyms——
-
PathwayOthers
-
TargetOther Targets
-
RecptorOthers
-
Research Area——
-
Indication——
Chemical Information
-
CAS Number28983-56-4
-
Formula Weight799.8
-
Molecular FormulaC37H27N3Na2O9S3
-
Purity>98% (HPLC)
-
SolubilityH2O:19 mg/mL?(23.75mM;?Need ultrasonic)
-
SMILESO=S(C1=CC=CC=C1/N=C2C=C/C(C=C\2)=C(C3=CC=C(NC4=CC=CC=C4S(=O)(O)=O)C=C3)\C5=CC=C(NC6=CC=CC=C6S(=O)([O-])=O)C=C5)([O-])=O.[Na+].[Na+]
-
Chemical Name——
Shipping & Storage Information
-
Storage(-20℃)
-
ShippingWith Ice Pack
-
Stability≥ 2 years
Reference
1.Mohammed Hassan, et al. Micellar effect on the kinetics of oxidation of methyl blue by Ce(IV) in sulfuric acid medium. Arabian Journal of Chemistry. Volume 8, Issue 1, January 2015, Pages 72-77.
molnova catalog



related products
-
CAY10465
CAY 10465 is a selective and high-affinity AhR agonist, with a Ki of 0.2 nM, and shows no effect on estrogen receptor (Ki >100000 nM).
-
[Sar1, Ile8]-Angiote...
[Sar1, Ile8]-Angiotensin II(3TFA) is a peptide that has multiple effects on vascular smooth muscle.
-
PPIase-Parvulin Inhi...
PPIase-Parvulin Inhibitor is a cell-pemeable inhibitor of the Pin1 and Pin4.



 Cart
Cart
 sales@molnova.com
sales@molnova.com


